Projects

Small cell lung carcinoma
SCLC is the most aggressive form of lung cancer with early systemic metastasis. Immune checkpoint blockade in combination with chemotherapy is approved for the treatment of advanced stage SCLC patients. However, immune checkpoint blockades are often not capable of achieving their effect in metastases. We therefore want to discover new targets in the metastatic cascade that can be therapeutically attacked.
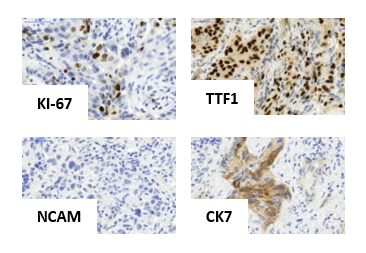
In immunohistochemistry (IHC), SCLC is indicated by a round and small cell morphology, a high proliferative index (KI-67), and frequent neuroendocrine cell-differentiation (NCAM, SYN).

Lung squamous cell carcinoma
The treatment of lung squamous cell carcinoma (LUSC) is challenging, as options for targeted therapy are very limited. For this reason, we aim to identify clinically relevant subgroups of LUSC that will allow novel therapy stratifications. Immune checkpoint blockade with combined additional treatments is a promising therapeutic approach and is likely to provide synergistic effects, which we will investigate.
LUSC frequently originates from basal cells lining the basement membrane and is therefore positively stained for cytokeratin 5 and 6 with IHC.
Lung adenocarcinoma
The lung adenocarcinoma (LUAD) is a subtype of cancer for which therapeutic options have made remarkable progress in recent years, as many vulnerable oncogenic genomic lesions have been identified. Unfortunately, there are still > 25 % of cases with no known genetic cause. For them, immune checkpoint blockade in combination with chemotherapy is an approved treatment. However, the tumors often exhibit initial or acquired resistance to therapy, the origin of these resistances has not been described in detail. We want to find a way to counteract resistance and enhance an anti-tumor immune response.
LUAD frequently expresses TTF1 and cytokeratin 7 in the IHC analysis and is classically non-neuroendocrine tumors.
Selected publications
ERBB2 signaling drives immune cell evasion and resistance against immunotherapy in small cell lung cancer. Meder L, Orschel CI, Bouchez CL, Gholamipoorfard R, Orschel CV, Stahl D, Kreer C, Koker M, Nill M, Kocak IG, Noh KW, Zhao X, Ullrich L, Häupl B, Jakob J, Eich ML, Florin A, Grüll H, Wolf J, Beleggia F, Büttner R, Oellerich T, Klein F, Brägelmann J, Reinhardt HC, Abedpour N, Ullrich RT. Nat Commun. 2025 Dec 9;16(1):10983. doi: 10.1038/s41467-025-66800-x. PMID: 41361170
Aberrations in the glycosylation of receptor tyrosine kinases: A focus on lung adenocarcinoma. Dmitrieva AM, Kocak IG, Meder L. Cytojournal. 2025 Jun 14;22:62. doi: 10.25259/Cytojournal_21_2025. eCollection 2025. PMID: 40708831
A Commentary on RIPK2: The Hidden Driver of MHC-I Related Immune Suppression in Cancer. Yang, Q., Ruff, A.L., Meder, L. (2025). BIOCELL, 49(2), 161–165. https://doi.org/10.32604/biocell.2025.060227
Blocking the angiopoietin-2-dependent integrin β-1 signaling axis abrogates small cell lung cancer invasion and metastasis. Meder L, Orschel CI, Otto CJ, Koker M, Brägelmann J, Ercanoglu MS, Dähling S, Compes A, Selenz C, Nill M, Dietlein F, Florin A, Eich ML, Borchmann S, Odenthal M, Blazquez R, Hilberg F, Klein F, Hallek M, Büttner R, Reinhardt HC, Ullrich RT.JCI Insight. 2024 May 22;9(10):e166402. doi: 10.1172/jci.insight.166402. PMID: 38775153
The role of LIN28B in tumor progression and metastasis in solid tumor entities. Gewalt T, Noh KW, Meder L. Oncol Res. 2023 Apr 10;31(2):101-115. doi: 10.32604/or.2023.028105. eCollection 2023. PMID: 37304235
EGFR Inhibition Strongly Modulates the Tumour Immune Microenvironment in EGFR-Driven Non-Small-Cell Lung Cancer. Selenz C, Compes A, Nill M, Borchmann S, Odenthal M, Florin A, Brägelmann J, Büttner R, Meder L, Ullrich RT. Cancers (Basel). 2022 Aug 16;14(16):3943. doi: 10.3390/cancers14163943. PMID: 36010935
Notch1 Deficiency Induces Tumor Cell Accumulation Inside the Bronchiolar Lumen and Increases TAZ Expression in an Autochthonous Kras LSL-G12V Driven Lung Cancer Mouse Model. Meder L, Florin A, Ozretić L, Nill M, Koker M, Meemboor S, Radtke F, Diehl L, Ullrich RT, Odenthal M, Büttner R, Heukamp LC. Pathol Oncol Res. 2021 Apr 16;27:596522. doi: 10.3389/pore.2021.596522. eCollection 2021. PMID: 34257546
Combined VEGF and PD-L1 Blockade Displays Synergistic Treatment Effects in an Autochthonous Mouse Model of Small Cell Lung Cancer. Meder L, Schuldt P, Thelen M, Schmitt A, Dietlein F, Klein S, Borchmann S, Wennhold K, Vlasic I, Oberbeck S, Riedel R, Florin A, Golfmann K, Schlößer HA, Odenthal M, Buettner R, Wolf J, Hallek M, Herling M, von Bergwelt-Baildon M, Reinhardt HC, Ullrich RT. Cancer Res. 2018 Aug 1;78(15):4270-4281. doi: 10.1158/0008-5472.CAN-17-2176. Epub 2018 May 18. PMID: 29776963
LIN28B enhanced tumorigenesis in an autochthonous KRASG12V-driven lung carcinoma mouse model. Meder L, König K, Dietlein F, Macheleidt I, Florin A, Ercanoglu MS, Rommerscheidt-Fuss U, Koker M, Schön G, Odenthal M, Klein F, Büttner R, Schulte JH, Heukamp LC, Ullrich RT. Oncogene. 2018 May;37(20):2746-2756. doi: 10.1038/s41388-018-0158-7. Epub 2018 Mar 5. PMID: 29503447
Notch signaling triggers the tumor heterogeneity of small cell lung cancer. Meder L, Büttner R, Odenthal M. J Thorac Dis. 2017 Dec;9(12):4884-4888. doi: 10.21037/jtd.2017.11.104. PMID: 29312683
Implementing amplicon-based next generation sequencing in the diagnosis of small cell lung carcinoma metastases. Meder L, König K, Fassunke J, Ozretić L, Wolf J, Merkelbach-Bruse S, Heukamp LC, Buettner R. Exp Mol Pathol. 2015 Dec;99(3):682-6. doi: 10.1016/j.yexmp.2015.11.002. Epub 2015 Nov 10. PMID: 26546837
NOTCH, ASCL1, p53 and RB alterations define an alternative pathway driving neuroendocrine and small cell lung carcinomas. Meder L, König K, Ozretić L, Schultheis AM, Ueckeroth F, Ade CP, Albus K, Boehm D, Rommerscheidt-Fuss U, Florin A, Buhl T, Hartmann W, Wolf J, Merkelbach-Bruse S, Eilers M, Perner S, Heukamp LC, Buettner R. Int J Cancer. 2016 Feb 15;138(4):927-38. doi: 10.1002/ijc.29835. Epub 2015 Sep 25. PMID: 26340530